SUIT-032 O2 mt D109: Difference between revisions
No edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
|info='''A''': protocol for simultaneous determination of O<sub>2</sub> flux and NADH autofluorescence in mitochondrial preparations (isolated mitochondria, tissue homogenate and permeabilized cells)- '''[[SUIT-006]]''' | |info='''A''': protocol for simultaneous determination of O<sub>2</sub> flux and NADH autofluorescence in mitochondrial preparations (isolated mitochondria, tissue homogenate and permeabilized cells)- '''[[SUIT-006]]''' | ||
|application=NADH | |application=NADH | ||
|SUIT number= | |SUIT number=D109_mt;1PGM;2D;3Myx.png | ||
}} | }} | ||
{{Template:SUIT text | {{Template:SUIT text D109}} | ||
__TOC__ | __TOC__ | ||
| Line 13: | Line 13: | ||
== Representative traces == | == Representative traces == | ||
[[File:SUIT-032 | [[File:SUIT-032 O2 mt D109 O2.png|600px]] | ||
{{Template:SUIT-032 | {{Template:SUIT-032 O2 mt D109}} | ||
== Strengths and limitations == | == Strengths and limitations == | ||
:::* SUIT-032 NADH mt D078 in combination with [[SUIT-033 NADH mt D081]] provides NAD redox ratios in LEAK and OXPHOS states, measured simultaneously with respiration. | :::* SUIT-032 NADH mt D078 in combination with [[SUIT-033 NADH mt D081]] provides NAD redox ratios in LEAK and OXPHOS states, measured simultaneously with respiration. | ||
| Line 35: | Line 34: | ||
== Chemicals and syringes == | == Chemicals and syringes == | ||
{{Template:Chemical SUIT-032 | {{Template:Chemical SUIT-032 O2 mt D109}} | ||
::: Suggested stock concentrations are shown in the specific DL-Protocol. | ::: Suggested stock concentrations are shown in the specific DL-Protocol. | ||
== References == | == References == | ||
{{#ask:[[Category:Publications]] [[Additional label::SUIT-032 | {{#ask:[[Category:Publications]] [[Additional label::SUIT-032 O2 mt D109]] [[Instrument and method::O2k-Protocol]] | ||
| ?Was published in year=Year | | ?Was published in year=Year | ||
| ?Has title=Reference | | ?Has title=Reference | ||
Revision as of 16:53, 4 January 2024
Description
Reference: A: protocol for simultaneous determination of O2 flux and NADH autofluorescence in mitochondrial preparations (isolated mitochondria, tissue homogenate and permeabilized cells)- SUIT-006
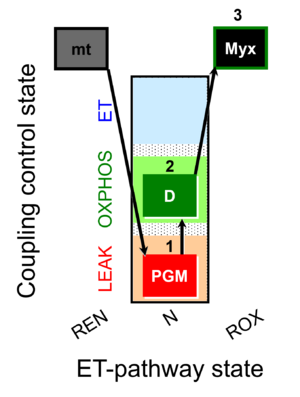
SUIT number: D109_mt;1PGM;2D;3Myx.png
O2k-Application: NADH
The coupling-control protocol SUIT-032 O2 mt D109 is used as a control protocol for SUIT-032 NADH mt D078, allowing for cytochrome c test and the evaluation of mitochondrial respiration in two coupling control states: LEAK, and OXPHOS in the N-pathway
After the addition of mitochondria in the absence of fuel substrates and ADP, Ren, respiration due to oxidation of endogenous substrates remaining after mitochondrial isolation is measured.
The addition of the complex III inhibitor myxothiazol allows the measurement of Rox.
Communicated by Grings M, Cardoso Luiza HD (last update 2023-12-21)
Representative traces
File:SUIT-032 O2 mt D109 O2.png
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| mt | REN | mt
| ||
| 1PGM | PGML(n) | N | CI | 1PGM
|
| 2D | PGMP | N | CI | 1PGM;2D
|
| 3Myx | ROX | 1PGM;2D;3Myx
|
- Bioblast links: SUIT protocols - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Coupling control
- Pathway control
- Main fuel substrates
- » Glutamate, G
- » Glycerophosphate, Gp
- » Malate, M
- » Octanoylcarnitine, Oct
- » Pyruvate, P
- » Succinate, S
- Main fuel substrates
- Glossary
Strengths and limitations
- SUIT-032 NADH mt D078 in combination with SUIT-033 NADH mt D081 provides NAD redox ratios in LEAK and OXPHOS states, measured simultaneously with respiration.
- + Reasonable duration of the experiment.
- + H2 gas from Oxia or N2/argon can be used to decrease O2 concentration to obtain anoxia faster.
- - Fully oxidized NAD can only be obtained with the combination with SUIT-033 NADH mt D081 or with samples in which endogenous substrates are absent.
- - Careful washing is required after the experiment to avoid carry-over of uncoupler and inhibitors. The addition of liver homogenate is recommended in the washing protocol to bind strong inhibitors.
- - The concentration of the oxidized and reduced NAD fraction cannot be determined.
- - Omy concentration has to be determined if used. Higher concentrations of Omy may inhibit the ET state.
- - Antimycin A and CCCP cannot be used due to the high chemical background effect on fluorescence.
- - Cytochrome c test cannot be performed during the protocol as it affects fluorescence. Cytochrome c test can be performed in the following protocol: SUIT-032 O2 mt D109.
- After myxothyazol titration, this protocol can be extended with the Complex IV assay.
Compare SUIT protocols
- SUIT-006 NADH mt D084: Protocol for simultaneous determination of O2 flux and NADH autofluorescence in isolated mitochondria. Similar protocol with uncoupler titrations and ET state evaluation.
- SUIT-033 NADH mt D081: Protocol for simultaneous determination of O2 flux and NADH autofluorescence in isolated mitochondria, allowing for calibration of the NAD redox ratios with samples that contain residual endogenous substrates. Additional titration of low concentration of ADP (0.1 μM) for depletion of endogenous substrates and calibration of fully reduced NAD, allowing for cross-calibration with SUIT-032 NADH mt D078.
- SUIT-032 O2 mt D109: Control protocol for respiration only, allowing for cytochrome c test.
Chemicals and syringes
If the experiment is performed as a control for D078, we recommend using the same chemicals.
| Step | Chemical(s) and link(s) | Comments |
|---|---|---|
| 1PGM | Pyruvate (P), Glutamate (G), and Malate (M) | |
| 2D | ADP (D) | |
| 3Myx | Myxothiazol |
- Suggested stock concentrations are shown in the specific DL-Protocol.