SUIT-003 AmR ce D017: Difference between revisions
Beno Marija (talk | contribs) No edit summary |
No edit summary |
||
| Line 15: | Line 15: | ||
__TOC__ | __TOC__ | ||
Communicated by [[Komlodi T]], [[Doerrier C]], | Communicated by [[Komlodi T]], [[Doerrier C]], [[Cardoso LHD]] (last update 2019-07-05) | ||
{{Template:SUIT-003 AmR ce D017}} | {{Template:SUIT-003 AmR ce D017}} | ||
Latest revision as of 00:09, 13 April 2021
Description
Abbreviation: CCP-ce S permeability test
Reference: B: CCP(P) and cell membrane permeability test with succinate - SUIT-003
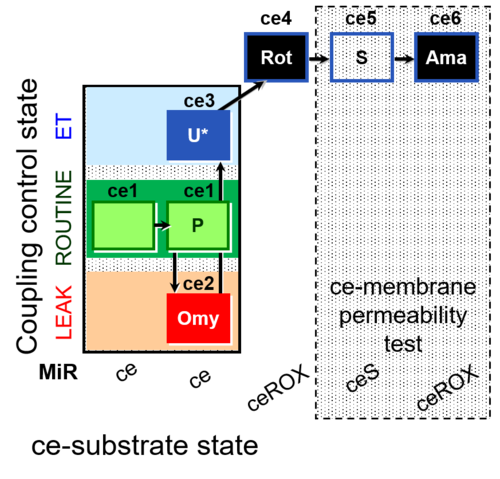
SUIT number: D017_ce1;ce2P;ce3Omy;ce4U;ce5Rot;ce6S;ce7Ama
O2k-Application: AmR
- MitoPedia: SUIT - coupling-control protocol (CCP) with pyruvate and cell membrane permeability test
- SUIT protocol pattern: ce1;ce1P;ce2Omy;ce3U;ce4Rot;ce5S;ce6Ama
This protocol has been designed to study the H2O2 production in various coupling control states and test the cell membrane permeability with the same protocol using living cells. Pyruvate is added to support the ROUTINE respiration of living cells when they are in a respiratory for mitochondrial preparations (i.e. MiR05). To initiate LEAK-respiration oligomycin is added, inhibiting the ATP-synthase and leading to an increased in the H2O2 production. Whereas titrations of uncoupler results in a decreased H2O2 generation. However, one of the main problems with living cells is that the H2O2 flux is very low or hardly detectable due to the scavenger activity of the antioxidant enzymes in the cytosol.
According to our results, the H2O2 production is linearly dependent on the O2 concentration in MiR05-Kit, therefore, during the measurements the O2 concentration has to be well-defined. In this protocol, we suggest not to go under ~100 µM O2. SUIT-018 AmR mt D041 has been designed to study O2 dependence of H2O2 flux in mitochondrial preparations.
Communicated by Komlodi T, Doerrier C, Cardoso LHD (last update 2019-07-05)
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 0DTPA |
| |||
| 0SOD |
| |||
| 0HRP |
| |||
| 0AmR |
|
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| ce1 | ROUTINE | ce1
| ||
| ce1P | ROUTINE | ce1;ce1P; ROUTINE, R
| ||
| ce2Omy | L(Omy) | ce1;ce1P;ce2Omy
| ||
| ce3U | E | ce1;ce1P;ce2Omy;ce3U
| ||
| ce4Rot | ROX | ce1;ce1P;ce2Omy;ce3U;ce4Rot
| ||
| ce5S | ce1;ce1P;ce2Omy;ce3U;ce4Rot;ce5S
| |||
| ce6Ama | ROX | ce1;ce1P;ce2Omy;ce3U;ce4Rot;ce5S;ce6Ama
|
Strengths and limitations
- When MiR05 is used, the addition of pyruvate is recommended to provide an external fuel substrate.
- When culture medium (e.g. DMEM) is used, application of phenol red is not advisable, since it can interfere with the fluorescence signal.
- + Reasonable duration of the experiments.
- - Minor changes in the H2O2 flux can be seen with living cells because of the action of the antioxidant enzymes in the cytosol, which scavenge the H2O2 produced. (AmR is not permeable to the cell membrane, therefore, only the H2O2 that crosses the plasma membrane can be measured)
Compare SUIT protocols
- SUIT-013 AmR ce D023: SUIT-013 has been designed to study the oxygen dependence of H2O2 production in intact cells.
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| MiPNet24.10 H2O2 flux analysis | 2021-10-22 | Hydrogen peroxide flux analysis using Amplex UltraRed assay in MiR05-Kit with DatLab 7.4 | ||
| Komlodi 2021 BEC AmR-O2 | 2021 | Komlódi T, Sobotka O, Gnaiger E (2021) Facts and artefacts on the oxygen dependence of hydrogen peroxide production using Amplex UltraRed. Bioenerg Commun 2021.4. https://doi.org/10.26124/bec:2021-0004 | Saccharomyces cerevisiae | Other cell lines |
| MiPNet20.14 AmplexRed H2O2-production | 2019-06-24 | O2k-FluoRespirometry: HRR and simultaneous determination of H2O2 production with Amplex UltraRed. | Mouse | Heart |
| Komlodi 2018 Methods Mol Biol | 2018 | Komlodi T, Sobotka O, Krumschnabel G, Bezuidenhout N, Hiller E, Doerrier C, Gnaiger E (2018) Comparison of mitochondrial incubation media for measurement of respiration and hydrogen peroxide production. Methods Mol Biol 1782:137-55. | Human Mouse | Skeletal muscle HEK |
| Makrecka-Kuka 2015 Biomolecules | 2015 | Makrecka-Kuka M, Krumschnabel G, Gnaiger E (2015) High-resolution respirometry for simultaneous measurement of oxygen and hydrogen peroxide fluxes in permeabilized cells, tissue homogenate and isolated mitochondria. https://doi.org/10.3390/biom5031319 | Human Mouse | Heart Nervous system HEK |
| Krumschnabel 2015 Methods Mol Biol | 2015 | Krumschnabel G, Fontana-Ayoub M, Sumbalova Z, Heidler J, Gauper K, Fasching M, Gnaiger E (2015) Simultaneous high-resolution measurement of mitochondrial respiration and hydrogen peroxide production. Methods Mol Biol 1264:245-61. | Mouse | Nervous system |
MitoPedia concepts:
SUIT protocol,
SUIT B,
Find
MitoPedia methods:
Fluorometry