Description
Abbreviation: NS(P)
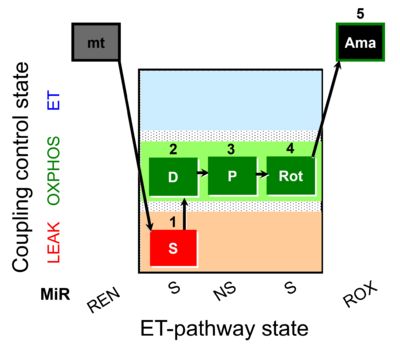
Reference: A: short protocol for H2O2 (AmR) in mitochondrial preparations; mt: isolated mitochondria, tissue homogenate and permeabilized cells - SUIT-009
SUIT number: D021_1S;2D;3P;4Rot;5Ama
O2k-Application: AmR
- MitoPedia: SUIT short protocol for H2O2 (AmR) for isolated mitochondria, tissue homogenate and permeabilized cells
- SUIT-category: NS(P)
- SUIT protocol pattern: orthogonal 1S;2D;3P;4Rot-
SUIT-009 is a short protocol for simultaneous determination of O2 flux and the rate of H2O2 production (H2O2 flux) in mitochondrial preparations such as isolated mitochondria, tissue homogenate (except of liver) and permeabilized cells (already permeabilized when they are added to the chamber). Succinate (S) supports the reverse electron transfer (RET)-related H2O2 production in the LEAK state. Pyruvate (P) supports NADH-pathway (N), and usually in the presence of ADP it does not increase further the H2O2 flux. Antimycin A (Ama) inhibits Complex III (CIII) at the Qo level and could increase the H2O2 flux. The sensitivity of the Amplex UltraRed® assay (for determining H2O2 production) changes over the experimental time and upon addition of chemicals. To correct H2O2 flux for the sensitivity changes several H2O2 calibration steps are done during the experiment. The experimental control for the O2-Application should be done with SUIT-009 O2 mt D015.
Communicated by Iglesias-Gonzalez J, Komlodi T and Gnaiger E (last update 2019-04-03)
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 0DTPA |
| |||
| 0SOD |
| |||
| 0HRP |
| |||
| 0AmR |
|
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 1S | SL(n) | S | CII | 1S
|
| 2D | SP | S | CII | 1S;2D
|
| 3P | NSP | NS | CI&II | 1S;2D;3P
|
| 4Rot | SP | S | CII | 1S;2D;3P;4Rot
|
| 5Ama | ROX | 1S;2D;3P;4Rot;5Ama
|
- Bioblast links: SUIT protocols - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Coupling control
- Pathway control
- Main fuel substrates
- » Glutamate, G
- » Glycerophosphate, Gp
- » Malate, M
- » Octanoylcarnitine, Oct
- » Pyruvate, P
- » Succinate, S
- Main fuel substrates
- Glossary
Strengths and limitations
- It is a protocol design to analyse:
- 1. the S- and NS-pathway linked respiration and ROS production.
- 2. any Complex I defect, which would lead to a decrease in the N-linked respiration and a reduced reverse electron transfer-supported ROS production.
- 3. any Complex II defect, which would result in an altered S-linked respiration and ROS production.
- SUIT-009 O2 mt D015: determination of O2 flux on isolated mitochondria, tissue homogenate and permeabilized cells (already permeabilized when they are added to the chamber) as a control for SUIT-009 AmR mt D021 to determine the inhibitory effect of the fluorescent dye on the mt-respiration.
- + This protocol is designed to evaluate RET in the S-linked pathway in the LEAK state, which might be inhibited by ADP due to the depolarisation of the mt-membrane.
- + Short protocol.
- - CIV activity and cytochrome c test cannot be performed together with the fluorescence assay.
- - We do not generally recommend the addition of already permeabilized cells into the chamber. Instead, we recommend performing the permeabilization of the plasma membrane into the chambers.
Compare SUIT protocols
- SUIT-006 AmR mt D048 to investigate the dependence of H2O2 flux on the mt-membrane potential on the N-control state in isolated mitochondria, tissue homogenate and permeabilized cells (already permeabilized when they are added to the chamber).
- SUIT-018 is a short protocol to study the oxygen dependence of O2 flux and H2O2 production on isolated mitochondria or tissue homogenate.
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| MiPNet24.10 H2O2 flux analysis | 2021-10-22 | Hydrogen peroxide flux analysis using Amplex UltraRed assay in MiR05-Kit with DatLab 7.4 | ||
| Komlodi 2021 BEC AmR-O2 | 2021 | Komlódi T, Sobotka O, Gnaiger E (2021) Facts and artefacts on the oxygen dependence of hydrogen peroxide production using Amplex UltraRed. Bioenerg Commun 2021.4. https://doi.org/10.26124/bec:2021-0004 | Saccharomyces cerevisiae | Other cell lines |
| MiPNet20.14 AmplexRed H2O2-production | 2019-06-24 | O2k-FluoRespirometry: HRR and simultaneous determination of H2O2 production with Amplex UltraRed. | Mouse | Heart |
| Komlodi 2018 Methods Mol Biol | 2018 | Komlodi T, Sobotka O, Krumschnabel G, Bezuidenhout N, Hiller E, Doerrier C, Gnaiger E (2018) Comparison of mitochondrial incubation media for measurement of respiration and hydrogen peroxide production. Methods Mol Biol 1782:137-55. | Human Mouse | Skeletal muscle HEK |
| Makrecka-Kuka 2015 Biomolecules | 2015 | Makrecka-Kuka M, Krumschnabel G, Gnaiger E (2015) High-resolution respirometry for simultaneous measurement of oxygen and hydrogen peroxide fluxes in permeabilized cells, tissue homogenate and isolated mitochondria. https://doi.org/10.3390/biom5031319 | Human Mouse | Heart Nervous system HEK |
| Krumschnabel 2015 Methods Mol Biol | 2015 | Krumschnabel G, Fontana-Ayoub M, Sumbalova Z, Heidler J, Gauper K, Fasching M, Gnaiger E (2015) Simultaneous high-resolution measurement of mitochondrial respiration and hydrogen peroxide production. Methods Mol Biol 1264:245-61. | Mouse | Nervous system |
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| Chang 2018 Life Sciences Meeting 2018 Innsbruck AT | 2018 | pH dependence of mitochondrial respiration and H2O2 production in oral cancer cells – a pilot study. | Human | HEK Kidney |
MitoPedia concepts:
SUIT protocol,
SUIT A,
Find
MitoPedia methods:
Fluorometry