Difference between revisions of "MiPNet08.16 pH calibration"
| Line 1: | Line 1: | ||
{{Publication | {{Publication | ||
|title=Gnaiger E | |title=Gnaiger E (2010) Laboratory protocol: pH measurement and temperature dependence of pH. Mitochondr Physiol Network 08.16(06): 1-5. | ||
|info=[[Media:MiPNet08.16 pH-Calibration.pdf|'''Open Access-MiPNet08.16.pdf''']]; [http://www.bioblast.at/index.php/File:MiPNet08.16_pH-Calibration.pdf Versions] [[Image:O2k-Protocols.jpg|right|150px|link=http://www.oroboros.at/?o2k-protocols|O2k-Protocols contents]] | |info=[[Media:MiPNet08.16 pH-Calibration.pdf|'''Open Access-MiPNet08.16.pdf''']]; [http://www.bioblast.at/index.php/File:MiPNet08.16_pH-Calibration.pdf Versions] [[Image:O2k-Protocols.jpg|right|150px|link=http://www.oroboros.at/?o2k-protocols|O2k-Protocols contents]] | ||
|authors=OROBOROS | |authors=OROBOROS | ||
|year= | |year=2010 | ||
|journal=Mitochondr Physiol Network | |journal=Mitochondr Physiol Network | ||
|abstract= | |abstract=pH of blood and intracellular compartments is tightly regulated. Blood pH at 37 °C is 7.4. However, when temperature is lowered, the pH of blood and of intracellular buffer systems increases, which has been reported as early as 1927 but was ignored for the following 30 years. The phenomenon of a change in blood pH with temperature of about -0.016 U/°C was rediscovered by comparative physiologists in studies of "cold blooded" vertebrates (turtles, fish), and is was recognized soon that the same pH/temperature relation applies to "warm blooded" mammals (rat, human). The physicochemical basis of pH/temperature relations and the consequences for acid-base balance and protein function were primarily analyzed by Rahn and Reeves (1979). Besides the importance for physiological and biochemical systems, the temperature dependence of pH of buffer systems has experimental significance for the measurement of pH at different temperatures, and for the choice of buffer systems when designing experiments at various temperatures. | ||
pH of blood and intracellular compartments is tightly regulated. Blood pH at 37 °C is 7.4. However, when temperature is lowered, the pH of blood and of intracellular buffer systems increases, which has been reported as early as 1927 but was ignored for the following 30 years. The phenomenon of a change in blood pH with temperature of about -0.016 U/°C was rediscovered by comparative physiologists in studies of "cold blooded" vertebrates (turtles, fish), and is was recognized soon that the same pH/temperature relation applies to "warm blooded" mammals (rat, human). The physicochemical basis of pH/temperature relations and the consequences for acid-base balance and protein function were primarily analyzed by Rahn and Reeves (1979). | |||
Besides the importance for physiological and biochemical systems, the temperature dependence of pH of buffer systems has experimental significance for the measurement of pH at different temperatures, and for the choice of buffer systems when designing experiments at various temperatures. | |||
:>> O2k-Protocols:[http://www.oroboros.at/?o2k-protocols Overall contents] | :>> O2k-Protocols:[http://www.oroboros.at/?o2k-protocols Overall contents] | ||
| Line 19: | Line 13: | ||
}} | }} | ||
{{Labeling | {{Labeling | ||
|area=Instruments;methods | |||
|topics=pH, Temperature | |topics=pH, Temperature | ||
|instruments=pH, Protocol | |instruments=pH, Protocol | ||
|additional=O2k-SOP | |additional=O2k-SOP | ||
}} | }} | ||
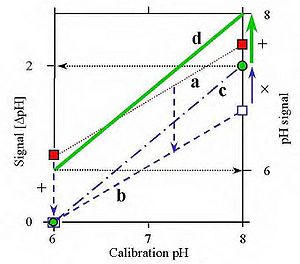
[[Image:MiPNet08.16.jpg|left|300px|thumb|Figure 1. Principle of a two-point calibration of a pH electrode system, at pH 6.00 and 8.00. (a) Signal before calibration (dotted line); (b) Calibration at pH 6 at electronic zero (0 mV), using the zero adjustment (+; additive as shown by the downwards arrows of equal length); (c) calibration of gain setting (slope) at pH 8, observing the pH of 2.0 units (x; multiplicative or proportional, as shown by the constant position of the 0 mV signal); (d) The full line of correspondence is obtained by shifting the entire curve to the correct absolute pH value (+, additive).]] | |||
== Further information == | == Further information == | ||
:>> [[O2k-pH ISE-Module]] | |||
>> [[PH_electrode|More details? Consult PH_electrode O2k-pH ISE-Module]] | :>> [[PH_electrode|More details? Consult PH_electrode O2k-pH ISE-Module]] | ||
In [http://www.oroboros.at/ | In [http://www.oroboros.at/?DatLab DatLab], the two-point calibration calculations are performed automatically (equations in [[MiPNet12.08]]). | ||
:>> [http://www.oroboros.at/?protocols_ph_calib Excel Templates and DatLab-Demo Files] | :>> [http://www.oroboros.at/?protocols_ph_calib Excel Templates and DatLab-Demo Files] | ||
== References == | |||
* Reeves RB, Rahn H (1979). Patterns in vertebrate acid-base regulation, Evolution of Respiratory processes: A Comparative Approach. Edited by Wood, S.C., Lenfant, C. New York, Marcel Dekker, Inc., pp 225-252. | |||
Revision as of 06:50, 27 February 2014
| Gnaiger E (2010) Laboratory protocol: pH measurement and temperature dependence of pH. Mitochondr Physiol Network 08.16(06): 1-5. |
» Open Access-MiPNet08.16.pdf; Versions
OROBOROS (2010) Mitochondr Physiol Network
Abstract: pH of blood and intracellular compartments is tightly regulated. Blood pH at 37 °C is 7.4. However, when temperature is lowered, the pH of blood and of intracellular buffer systems increases, which has been reported as early as 1927 but was ignored for the following 30 years. The phenomenon of a change in blood pH with temperature of about -0.016 U/°C was rediscovered by comparative physiologists in studies of "cold blooded" vertebrates (turtles, fish), and is was recognized soon that the same pH/temperature relation applies to "warm blooded" mammals (rat, human). The physicochemical basis of pH/temperature relations and the consequences for acid-base balance and protein function were primarily analyzed by Rahn and Reeves (1979). Besides the importance for physiological and biochemical systems, the temperature dependence of pH of buffer systems has experimental significance for the measurement of pH at different temperatures, and for the choice of buffer systems when designing experiments at various temperatures.
- >> O2k-Protocols:Overall contents
- >> Product: OROBOROS Oxygraph-2k, O2k-Catalogue
• Keywords: HRR-MultiSensor
• O2k-Network Lab: AT_Innsbruck_OROBOROS
Labels: MiParea: Instruments;methods
Regulation: pH, Temperature
HRR: pH, Protocol"Protocol" is not in the list (Oxygraph-2k, TIP2k, O2k-Fluorometer, pH, NO, TPP, Ca, O2k-Spectrophotometer, O2k-Manual, O2k-Protocol, ...) of allowed values for the "Instrument and method" property.
O2k-SOP
Further information
In DatLab, the two-point calibration calculations are performed automatically (equations in MiPNet12.08).
References
- Reeves RB, Rahn H (1979). Patterns in vertebrate acid-base regulation, Evolution of Respiratory processes: A Comparative Approach. Edited by Wood, S.C., Lenfant, C. New York, Marcel Dekker, Inc., pp 225-252.

