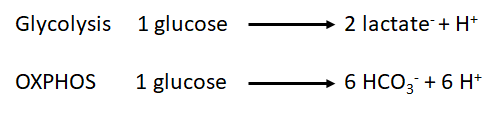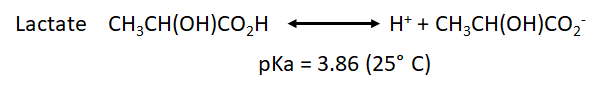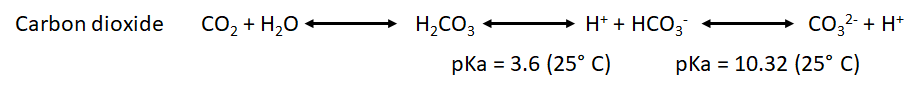Difference between revisions of "Talk:Hydrogenion flux"
m |
|||
| (29 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
__TOC__ | |||
== For critical evaluation == | |||
:::: The measurement of H<sup>+</sup> flux alone is not sufficient to determine, if the origin of H<sup>+</sup> is the glycolysis or other sources. For example, the carbon dioxide formed during the mitochondrial respiration acts as a net source of H<sup>+</sup> into the media and as consequence has to be taken into account. During the oxidation of the glucose, we have two main metabolic pathways involved and both have a net effect over the H<sup>+</sup> flux: | |||
:::: H2CO3 is not fully dissociated. Therefore, there is not a simple and constant stoichiometry between bicarbonate and H<sup>+</sup> production. | |||
| | [[File:Proton production Metabolic pathways.png|center|400ppx]] | ||
:[[File:Lactate.png|center|400ppx]] | |||
:[[File:Carbon dioxide.png|center|400ppx]] | |||
== Proton flux versus ECAR == | == Proton flux versus ECAR == | ||
The extracellular acidification rate (ECAR) is the change of pH in the incubation medium over time and can only be measured in a closed system. pH is the negative decadic logarithm of proton activity which is, in diluted solutions, in close approximation to the negative decadic logarithm of proton concentration. | :::: The extracellular acidification rate (ECAR) is the change of pH in the incubation medium over time and can only be measured in a closed system (why?). pH is the negative decadic logarithm of proton activity which is, in diluted solutions, in close approximation to the negative decadic logarithm of proton concentration. | ||
:::: Thus, measured changes in pH over time (ECAR) must be transformed from the logarithmic to the linear scale to obtain proton flux. | |||
Therefore, ECAR is of interest in relation to acidification issues in the incubation buffer or culture medium but must not be confused with the physiologically relevant metabolic proton flux. | Therefore, ECAR is of interest in relation to acidification issues in the incubation buffer or culture medium but must not be confused with the physiologically relevant metabolic proton flux. | ||
== | == H<sup>+</sup> flux and glycolysis == | ||
Measurement of | :::: Measurement of proton flux and glycolysis are related under specifically controlled conditions. Such conditions must be carefully evaluated, may require modifications of protocols, must be corrected for acid-base reactions unrelated to glycolysis and thus need data analysis beyond reporting changes of pH. | ||
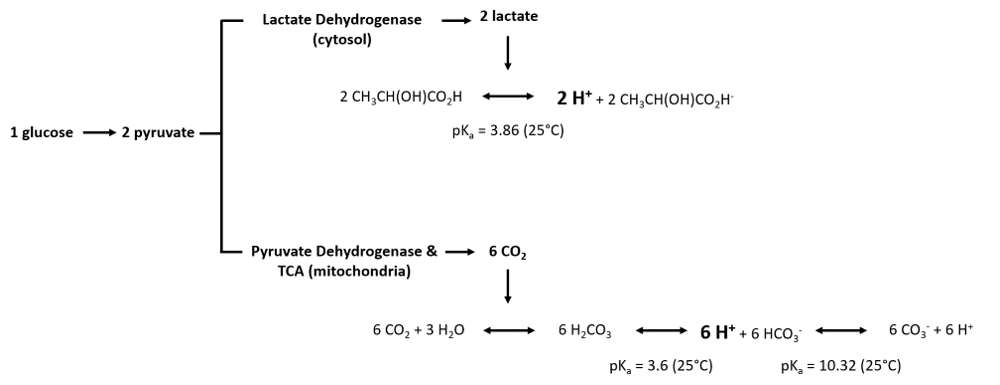
:::: Glycolysis is the degradation of glucose to pyruvate. Depending on the subsequent metabolism of pyruvate, glycolysis is indirectly related to H<sup>+</sup> flux. Pyruvate can either be converted to lactate, catalyzed by [[lactate dehydrogenase]] in the cytosol, or converted to Acetyl-CoA catalysed by [[pyruvate dehydrogenase]]), feeding into the [[Tricarboxylic acid cycle | TCA cycle]] (mitochondria). The catabolism of pyruvate can have an impact on proton flux as illustrated by the following equations: | |||
[[File:Proton_production.png]] | |||
As we can observe, the production of protons per molecule of glucose is three times higher in the mitochondria via dissolution of carbone dioxide than by conversion of pyruvate to lactate in the cytosol. Furthermore, the equations illustrate that the measurement of proton flux alone is not sufficient to determine the origin of protons. Experimental settings can help to estimate the main source of proton production, e.g. by inhibition of [[Oxidative phosphorylation | OXPHOS]] (link to SUIT protocols). | |||
To accurately measure proton flux induced by biological sample, the buffering capacity of the medium has to be small but still sufficient to keep the pH in the desired range for a limited period of time. Furthermore, the buffering capacity has to be determined ([[MiPNet23.15 O2k-pH ISE-Module]]) and taken into account when proton flux is calculated from the measured changes in pH. An alternative approach is to use buffers with very low buffering capacity and keep the pH value inside the desired limits by a [[O2k-pH_ISE-Module#pH-Stat|pH-Stat]]. Here, proton flux can be calculated either by changes in pH over time (previous calculation of buffering capacity of the medium required) or by the amount of injected base via pH Stat. [[MiPNet23.15 O2k-pH ISE-Module]] | |||
== Measurement of proton flux with the O2k-pH ISE-Module == | == Measurement of proton flux with the O2k-pH ISE-Module == | ||
:::: The [[Oroboros O2k]] supports the modular O2k-MultiSensor extension for recording potentiometric (voltage) signals simultaneously with the oxygen signals in both O2k-chambers. Potentiometric measurements result in a voltage signal ('''pX''') which is typically a linear function of the logarithm of the activity (concentration) of the substance of interest (the ''analyte''). A calibrated pH electrode displays the negative decadic logarithm of the H<sup>+</sup> ion activity (potentia hydrogenii) and thus got its name “pH electrode”. | :::: The [[Oroboros O2k]] supports the modular O2k-MultiSensor extension for recording potentiometric (voltage) signals simultaneously with the oxygen signals in both O2k-chambers. Potentiometric measurements result in a voltage signal ('''pX''') which is typically a linear function of the logarithm of the activity (concentration) of the substance of interest (the ''analyte''). A calibrated pH electrode displays the negative decadic logarithm of the H<sup>+</sup> ion activity (potentia hydrogenii) and thus got its name “pH electrode”. | ||
== Applications == | |||
:::: The majority of novel applications will address aerobic or anaerobic glycolysis in intact cells, using the measurement of proton flux as an indirect but continuous record of lactate production and corresponding acidification of the medium, while simultaneously monitoring oxygen concentration and oxygen consumption. For this application, specific experimental settings are required to leave lactic acid production as the dominant mechanism of acidification. | |||
:::: The pH electrode in the O2k can also be used in conjunction with a study of mitochondrial permeability transition (e.g. [[SE_Lund_Elmer E]]). | |||
:::: For simultaneous measurement of O<sub>2</sub> and pH, we refer to the classical literature on bioenergetics and the discovery of the chemiosmotic coupling mechanism, the quantification of H<sup>+</sup>/O<sub>2</sub> stoichiometric ratios for proton pumping (Peter Mitchell). | |||
== O2k signal and output == | == O2k signal and output == | ||
:::# [[O2k signals and output#Signal of the O2k and add-on modules |O2k signal]]: The [[O2k-pH ISE-Module]] is operated through the pX channel of the O2k, with electric potential (volt [V]) as the primary and raw signal | :::# [[O2k signals and output#Signal of the O2k and add-on modules |O2k signal]]: The [[O2k-pH ISE-Module]] is operated through the pX channel of the O2k, with electric potential (volt [V]) as the primary and raw signal | ||
| Line 57: | Line 50: | ||
== Compare measurement of pH with the pH electrode and ratiometric fluorometric methods == | |||
== Compare measurement of pH with the pH electrode and ratiometric fluorometric methods | |||
::::» [[Carboxy SNARF 1]] | ::::» [[Carboxy SNARF 1]] | ||
::::» [[HPTS]] | ::::» [[HPTS]] | ||
{{Keywords: pH}} | |||
== References == | |||
::::* [[MiPNet08.16 pH calibration]] | |||
::::* [[MiPNet15.03 O2k-MultiSensor-ISE]] | |||
::::* [[MiPNet23.15 O2k-pH ISE-Module]] | |||
:::: | ::::* [[MiPNet24.06 Oxygen flux analysis - DatLab 7.4]] | ||
:::: | ::::* [[MiPNet12.10 TIP2k-manual]] | ||
::::* [[MiPNet15.08 TPP electrode]] | |||
::::* [[MiPNet22.11 O2k-FluoRespirometer manual]] | |||
»{{MitoPedia O2k and high-resolution respirometry | |||
|mitopedia O2k and high-resolution respirometry=O2k hardware | |||
}} | }} | ||
»{{MitoPedia O2k and high-resolution respirometry | |||
|mitopedia O2k and high-resolution respirometry=DatLab | |||
}} | }} | ||
{{Technical support integrated}} | |||
{{MitoPedia concepts | {{MitoPedia concepts | ||
| Line 113: | Line 87: | ||
}} | }} | ||
{{MitoPedia methods | {{MitoPedia methods | ||
|mitopedia method=Respirometry | |mitopedia method=Respirometry | ||
}} | }} | ||
Latest revision as of 14:04, 5 December 2020
For critical evaluation
- The measurement of H+ flux alone is not sufficient to determine, if the origin of H+ is the glycolysis or other sources. For example, the carbon dioxide formed during the mitochondrial respiration acts as a net source of H+ into the media and as consequence has to be taken into account. During the oxidation of the glucose, we have two main metabolic pathways involved and both have a net effect over the H+ flux:
- H2CO3 is not fully dissociated. Therefore, there is not a simple and constant stoichiometry between bicarbonate and H+ production.
Proton flux versus ECAR
- The extracellular acidification rate (ECAR) is the change of pH in the incubation medium over time and can only be measured in a closed system (why?). pH is the negative decadic logarithm of proton activity which is, in diluted solutions, in close approximation to the negative decadic logarithm of proton concentration.
- Thus, measured changes in pH over time (ECAR) must be transformed from the logarithmic to the linear scale to obtain proton flux.
Therefore, ECAR is of interest in relation to acidification issues in the incubation buffer or culture medium but must not be confused with the physiologically relevant metabolic proton flux.
H+ flux and glycolysis
- Measurement of proton flux and glycolysis are related under specifically controlled conditions. Such conditions must be carefully evaluated, may require modifications of protocols, must be corrected for acid-base reactions unrelated to glycolysis and thus need data analysis beyond reporting changes of pH.
- Glycolysis is the degradation of glucose to pyruvate. Depending on the subsequent metabolism of pyruvate, glycolysis is indirectly related to H+ flux. Pyruvate can either be converted to lactate, catalyzed by lactate dehydrogenase in the cytosol, or converted to Acetyl-CoA catalysed by pyruvate dehydrogenase), feeding into the TCA cycle (mitochondria). The catabolism of pyruvate can have an impact on proton flux as illustrated by the following equations:
As we can observe, the production of protons per molecule of glucose is three times higher in the mitochondria via dissolution of carbone dioxide than by conversion of pyruvate to lactate in the cytosol. Furthermore, the equations illustrate that the measurement of proton flux alone is not sufficient to determine the origin of protons. Experimental settings can help to estimate the main source of proton production, e.g. by inhibition of OXPHOS (link to SUIT protocols).
To accurately measure proton flux induced by biological sample, the buffering capacity of the medium has to be small but still sufficient to keep the pH in the desired range for a limited period of time. Furthermore, the buffering capacity has to be determined (MiPNet23.15 O2k-pH ISE-Module) and taken into account when proton flux is calculated from the measured changes in pH. An alternative approach is to use buffers with very low buffering capacity and keep the pH value inside the desired limits by a pH-Stat. Here, proton flux can be calculated either by changes in pH over time (previous calculation of buffering capacity of the medium required) or by the amount of injected base via pH Stat. MiPNet23.15 O2k-pH ISE-Module
Measurement of proton flux with the O2k-pH ISE-Module
- The Oroboros O2k supports the modular O2k-MultiSensor extension for recording potentiometric (voltage) signals simultaneously with the oxygen signals in both O2k-chambers. Potentiometric measurements result in a voltage signal (pX) which is typically a linear function of the logarithm of the activity (concentration) of the substance of interest (the analyte). A calibrated pH electrode displays the negative decadic logarithm of the H+ ion activity (potentia hydrogenii) and thus got its name “pH electrode”.
Applications
- The majority of novel applications will address aerobic or anaerobic glycolysis in intact cells, using the measurement of proton flux as an indirect but continuous record of lactate production and corresponding acidification of the medium, while simultaneously monitoring oxygen concentration and oxygen consumption. For this application, specific experimental settings are required to leave lactic acid production as the dominant mechanism of acidification.
- The pH electrode in the O2k can also be used in conjunction with a study of mitochondrial permeability transition (e.g. SE_Lund_Elmer E).
- For simultaneous measurement of O2 and pH, we refer to the classical literature on bioenergetics and the discovery of the chemiosmotic coupling mechanism, the quantification of H+/O2 stoichiometric ratios for proton pumping (Peter Mitchell).
O2k signal and output
- O2k signal: The O2k-pH ISE-Module is operated through the pX channel of the O2k, with electric potential (volt [V]) as the primary and raw signal
- O2k output: type I and II
Compare measurement of pH with the pH electrode and ratiometric fluorometric methods
- » Carboxy SNARF 1
- » HPTS
- Bioblast links: pH and protons - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- pH and protons
- » pH
- » hydrogen ion H+
- » hydron H+
- » hydronium ion H3O+
- » hydride H-
- » proton p+
- » pH buffering capacity
- » proton flux
- » proton pump versus hydrogen ion pump
- » proton leak
- » proton slip
- » protonmotive force
- pH and protons
- O2k-pH
- » O2k-Catalogue: O2k-pH ISE-Module
- » O2k-Manual pH electrode: MiPNet23.15 O2k-pH ISE-Module
- » O2k-SOP: MiPNet08.16 pH calibration
- » File:PH-Calibration-List.xls
- » NextGen-O2k, ratiometric: Carboxy SNARF 1
- » NextGen-O2k, ratiometric: HPTS
- » pH calibration buffers
- O2k-pH
- O2k-Publications
- HRFR - general
- » O2k-Manual: MiPNet22.11 O2k-FluoRespirometer manual
- » O2k signals and output
- » O2k-SOP: MiPNet14.06 Instrumental O2 background
- » MiPNet19.18A O2k-Series G: Start
- » ESD
- » O2k configuration
- » O2k control
- » O2k-FluoRespirometer
- » O2k-Main Unit#O2k-Series
- » Titration-Injection microPump
- » Compare: O2k-TPP+_ISE-Module
- HRFR - general
- DatLab
References
» MitoPedia O2k and high-resolution respirometry: O2k hardware
» MitoPedia O2k and high-resolution respirometry: DatLab
MitoPedia O2k and high-resolution respirometry:
O2k-Open Support
MitoPedia concepts: MiP concept
MitoPedia methods:
Respirometry